|
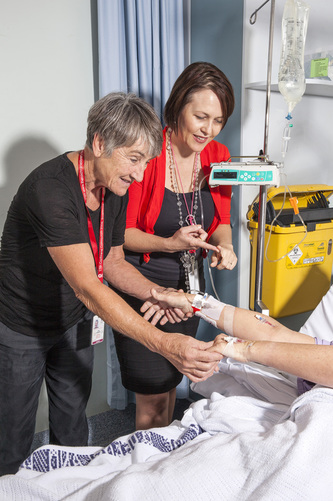
Dr. Claire Rickard is a PhD nurse investigator affiliated with Griffith University in Brisbane, Australia. An accomplished researcher who has published in top-tier journals and is internationally recognized as a leader in vascular access, she also oversees the Alliance for Vascular Alliance Teaching and Research Group (known widely as AVATAR). AVATAR and it's members have conducted some of the most influential trials in vascular access, including the recent routine vs. clinically warranted replacement of peripheral IVs (PIV) published in the Lancet.
Dr. Rickard was kind enough to sit down with us and answers some questions about vascular access and her research in this field. She has filled the ICJME form and has disclosed the following conflicts of interest: grants and other from 3M, grants and other from BBRAUN, grants and other from BD, grants from CAREFUSION, grants from COOK, grants from ENTROTECH, grants from BAXTER, grants from HOSPIRA, other from BARD, grants and other from TELEFLEX, outside the submitted work. VC: Please tell us a bit about yourself and how you got interested in vascular access? CR: I’m a Registered Nurse from Brisbane, Australia. I worked in medical and cardiac surgical wards (floors) and then in ICU for many years. I was always intrigued about all the variation in how we cared for vascular access devices between different hospitals. So, later I did (was almost forced to!) complete a research methods subject as I completed an ICU Graduate Diploma. It wasn' too longer afterwards that I could see how research just might be a good tool to sort out some of that care variation. And the rest, as they say, is history VC: Your research group, AVATAR, is well-known for producing high quality research. How did the group come to be and tell us a bit about your focus? CR: AVATAR came about quite organically over 20 years. When I say organically, I mean that it just sort of happened from the ground up - there was never some type of "master plan" to develop a research group focused on vascular access. At first it was just me and a couple of colleagues who shared this passion and interest. But then, as we were able to win larger research grants we met other researchers passionate about vascular access and just enjoyed learning from and spurring each other on. And so -AVATAR was born. Now keep in mind, AVATAR is a voluntary group - we don’t receive any ongoing funding from any of the hospitals or universities. But somehow we have managed to survive and thrive - speaking to the power of passionate individuals coming together to advance science that they truly care about. VC: Tell us about some of your ongoing research projects, especially those that relate to PICCs? CR: We have about 70 research projects active right at this minute. Our bigger PICC projects at the moment are the RSVP Trial which is randomising patients to have 4 day or 7 day routine replacement of their IV tubing, and the PISCES Trial which is another multi-centre randomised trial comparing methods of dressing and securement that prevent infection, dislodgment and thrombosis. VC: Is much of AVATARs research has focused on peripheral IVs? If so, can you share with us why you chose this device in particular? CR: Well actually this is a common misconception. As a former ICU nurse (in Australia we rarely see PIVs in ICU) I was initially far more focussed on central and arterial device issues. Later on, I could see the commonality of the problems across all vascular access devices, and realised that although there were pockets of research activity in central lines in particular, the poor old PIVs were almost totally ignored. Looking at the bigger picture made me realise that improving PIV care was actually going to benefit a huge majority of the population, due to the numbers used. VC: What have some of the surprises been in your research journey? CR: Well in Australia until recently we had no vascular access society (now we have avas.org.au), and IV/PICC teams continue to be rare, although brilliant when we have them. So I was actually unaware that there was this whole specialty out there called vascular access or infusion nursing until many years after I started research in the area. Now I’m so happy and energised to be connected with this international community of practitioners and researchers. The other thing that surprises me constantly is how content healthcare professionals can be with using and implementing new devices and care procedures, that have little to zero evidence that they actually work. That’s very frustrating. VC: Your team focuses on systematic reviews and trials? Why these two types of research methods? CR: Well we only have so many hours in this life! and we know that we need ‘Level 1 Evidence’, like Cochrane systematic reviews, and randomised clinical trials, if we want to understand ‘cause and effect’ relationships and identify those policies and products that really do prevent complications. It’s hard yakka (that's Australian for hard work btw) to do research so we think we should focus our limited time and money on providing the most valid and reliable research results for implementing in practice. VC: What advice do you have for vascular access nurses interested in starting a research project? CR: I would say go for it! But in reality you’re going to need some friends in the form of supportive clinical colleagues, and some experienced researchers who you can go to for advice. They don’t need to be vascular access researchers necessarily, you might have clinical trial nurses attached to the cancer or renal service for example, who would have heaps of knowledge about all the processes and paperwork. It would also help to find a mentor, either an academic nurse or doctor, but check they have published clinically focussed research themselves. I’d always suggest doing a randomised trial, not a pre and post study. It is actually no harder and your results will have so much more power. Even if you study a small number of patients, it’s very valuable data and can also then be included by authors of systematic reviews on the topic. VC: You’ve discussed creating a “Center of Excellence” for vascular access. Tell us a bit more about this. CR: We’d like to formalise what we have been doing in AVATAR for some time now. We are seeking government and philanthropic funds that would give us 5 years surety of funding. This money allows us to hire clinical nurses or doctors as researchers, so they can get that experience, and similarly fund Masters and PhD research scholarships so we can grow the global capacity for research. We plan to focus these research programs in three areas: (i) Insertion (assessment, procedure, devices), (ii) the Function phase (dressings and securement, flushing and blood sampling, connectors), and (iii) Removal (this one is a no-brainer - ongoing assessment and getting devices out when no longer needed). Some of this work will be clinical research but a whole other arm will focus on the Implementation/uptake of knowledge from research results. VC: In your opinion, what are the three most important questions remaining to be answered in vascular access? How can we go about getting these addressed? What barriers, if any, remain in effectively answering these issues? CR: Wow, ok. Here we go: #1: We know 25-50% of vascular access devices have complications that lead to the device becoming non-functional, but how do we set up public benchmarking, so that hospitals take this seriously, and practitioners are more aware of this? We plan to set up a Vascular Access Registry to help with this, but that will still need to find ways to be championed throughout the Quality, Nursing and Hospitalist worlds in particular, to make this successful. #2: In recent years, our research has shown that PIVs do not need to be routinely removed every 72-96 ours. They are licensed for up to 30 days use and with continued policies of clinical monitoring, can be safely used until there is a clinical reason for removal. We also know thanks to Dr Chopra and colleagues that PICCs have complications more similar to CVCs, so we are now taking that decision to place a PICC more seriously. So there is now this huge ‘middle ground’ – which patients/treatments can we use PIVs for (including new longer PIVs and mid-lines), and avoid the need for a PICC? #3. Occlusion is the big ticket item, it’s the number one complication across most device types, not phlebitis or infection as many people think. In terms of picking the ‘low hanging fruit’ – what can we do with better care practices ?flushing ?dressings ?connectors etc to help there. If we could even bring occlusion down by 10% in PIV and central lines, that would be a massive cost saving and patient benefit across the system. The barriers are simple: lack of funded research nurse positions within the health services, and lack of awareness across the broader health system about the catastrophe of vascular access complications occurring every day. VC: If there were one thing you wish you could have done differently in your research journey, who would it have been and why? CR: I would have perhaps done less individual research studies and spent more time on promoting implementation of results. It’s a tough call though, as I always have a lot of exciting new ideas I want to pursue. A decade ago, we thought once we’d published, as researchers our job was done and we could move on to the next topic. Now, we know it takes a lot of effort to get the health system to change and take on new policies. I’m not saying it’s necessarily the researcher’s job to do this, but they can help a lot as the various societies update their guidelines for example. 9/25/2023 03:12:25 am
What an inspiring interview with Dr. Claire Rickard! Her journey into the world of vascular access research is a testament to the power of curiosity and the impact of passionate individuals in the field of healthcare. Thank you for sharing this insightful interview with Dr. Claire Rickard. Her dedication to advancing vascular access care is truly commendable, and her work is making a meaningful difference in the field of healthcare. Comments are closed.
|
AuthorsBlogs written and edited by Vineet Chopra unless otherwise stated in the header. Guest blogs are identified accordingly. Archives
May 2019
Categories |